Abstract
Introduction: We frequently observe differences between results obtained in clinical trials and real-life. Several reasons may account for these discrepancies: age and coexisting conditions (older patients with additional comorbidities are often excluded from clinical trials), drug interactions and change of schedule (dose-intensity) as compared to the one used in the clinical trials. Azacitidine (AZA) combined with venetoclax (VEN) became the standard of care for acute myeloid leukemia (AML) for patients deemed ineligible for intensive chemotherapy. Composite complete remission (cCR) was 66.4% with median overall survival at 14.7 months (DiNardo et al., NEJM 2020). Recent real-life studies reported less promising results showing cCR between 40-70% and median overall survival (OS) between 7-12 months (Winters et al. Blood 2019; Garciaz et al. EHA 2022; Lam et al. EHA 2022). We report in this study the results of a single center cohort of AML patients receiving an AZA + VEN induction cycle strictly similar to the one reported in the VIALE-A clinical trial without drug interaction.
Methods: All patients were treated using VIALE-A guidelines. The first cycle consisted in AZA 75/mg/m² D1-7 + VEN 100mg D1, 200 mg D2, 400 mg D3-28. Treatment was delivered to inpatients, patients were discharged when complete remission was obtained with ANC recovery. All patients received antifungal prophylaxis by liposomal amphotericin B 3mg/kg 3 times a week during the first cycle without VEN dose reduction. For patients who achieved complete remission after cycle 1, the dose of VEN was reduced in case of grade 4 neutropenia and cycle 2 was delayed. For non-responders' patients, cycle 2 started straight after the end of cycle 1. Response was evaluated according to the 2017 European Leukemia Network (ELN) criteria. OS was calculated from the time of diagnosis of AML to the time of death or last known follow-up. Median OS was estimated using the Kaplan−Meier method, and differences between subgroups were assessed using the log-rank test.
Results:
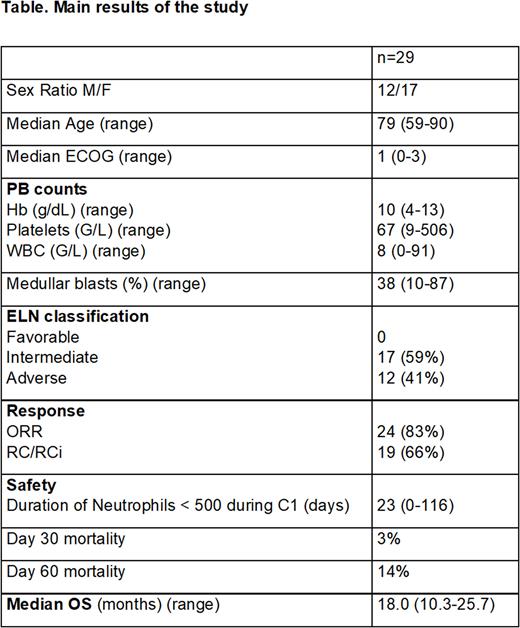
Twenty-nine (n=29) de novo AML patients were enrolled in this study in University Hospital of Nice. The median age was 79 years [range, 59-90] , Sex ratio M/F was 12/17 and median ECOG was 1 [range,0-3]. ELN 2017 prognostic classification was intermediate in 17 (59%) and adverse in 12 (41%) of patients. The most frequent mutations were NPM1, TP53 and TET2 in 6 (21%), ASXL1 in 5 (17%), DNMT3A in 3 (10%) patients, Median number of AZA + VEN cycles was 2 [range, 1-15]. Each patients of this study (100%) received VEN 400mg/d D1-28 during the first cycle without drug interactions needing VEN dose reduction.
Twenty four of the 29 patients (83%) of patients received liposomal amphotericin B prophylaxis during the first cycle. Seven of the 29 patients (24%) patients developed a possible or probable aspergillosis (5 possible, 2 probable). Median duration of neutropenia was 23 days [range,0-116]. Febrile neutropenia occurred in 27/29 (93%) of patients.
The overall response rate was 83% including 66% of cCR. Response was obtained after one cycle for 21 patients and after two cycles for 3 patients. Day-30 mortality was 3% and day-60 mortality was 14%. Median OS was 18 months [10.3-25.7, CI 95%].
Conclusions: Our data revealed that a strict introduction of AZA + VEN and a special attention in the monitoring for elderly patients with AML, appears feasible in real-life and was associated to similar outcomes than those observed in the VIALE-A study.
Liposomal amphotericin B prophylaxis was also feasible without any need of VEN dose reduction and led to improvements. These outcomes need to be confirmed in a larger cohort.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.